�Ѵ��ļ���photo/202083192141889.png
Research Background
Metal-organic frameworks (MOFs) are a class of highly ordered crystalline porous coordination polymers (PCPs). MOFs have the advantages of clear structure, high specific surface area and porosity, adjustable pore size, and easy chemical functionalization. They are considered to be a class of promising nano drug carriers. In recent years, the research of metal-organic framework (MOF) for biomedicine has attracted much attention.
Metal�COrganic Framework Nanocarriers for Drug Delivery in Biomedical ApplicationsYujia Sun, Liwei Zheng, Yu Yang, Xu Qian, Ting Fu*, Xiaowei Li, Zunyi Yang, He Yan, Cheng Cui*, Weihong Tan* Nano‑Micro Lett. (2020) 12:103https://doi.org/10.1007/s40820-020-00423-3
Highlights of this article
1. The application progress of metal organic framework (MOF) nano-medicine carriers in the field of biomedicine is reviewed. 2. The latest strategies for functionalization of therapeutic MOFs and their advantages and disadvantages are reviewed.
brief introduction
In this article, the team of Molecular Science and Biomedical Laboratory (MBL) of Hunan University reviewed the unique properties of MOFs and their applications as nano-drug carriers in the biomedical field. The current research status of the functionalization of MOFs using surface adsorption, pore encapsulation, covalent bonding, and functional molecules as building elements are reviewed. Subsequently, the latest biological applications of MOFs in the intracellular delivery of drugs, proteins and nucleic acids, especially nucleic acid aptamers, are introduced. Finally, this article has a comprehensive discussion on the challenges and prospects it faces, and provides a background for the future development of MOFs as efficient drug delivery systems.
Graphic guide
Introduction to I MOFs and its application in biomedicine
Metal-organic frameworks (MOFs) take inorganic metal (such as transition metals and lanthanide metals) ions/clusters as nodes and are supported by organic ligands (such as carboxylates, phosphonates, imidazolates and phenolic acid esters) , Forming an extended network of infinite 1D/2D/3D MOFs. In recent years, the application of MOFs in the biomedical field has attracted more and more attention. When the size of MOF particles is reduced to the nanometer level, these nano-MOFs (NMOFs) can be used as drug delivery vehicles for imaging, chemotherapy, photothermal therapy, or photodynamic therapy. Compared with other porous materials, MOFs have many outstanding advantages, such as: (1) It has a high specific surface area and porosity, and can be used for high loading therapeutic drugs; (2) It is easy to pass inorganic clusters and/or organic compounds. The body modifies the physical (such as pore size and shape) and chemical properties of MOFs. In addition, through the pre-design of the ligand or the modification method after synthesis, the required functional groups can be added to the organic ligand; (3) The open windows and pores of MOFs can make the diffusion matrix and the binding molecule interact; 4) Medium-strength coordination bonds make MOF biodegradable; (5) A clear structure is conducive to the study of host-guest interactions. Due to these unique properties, MOFs are considered to be one of the best drug candidates for drug delivery and cancer treatment. II Functionalized MOFs for drug delivery have unique properties, such as highly ordered structure, high specific surface area and large pore volume, which enable them to adsorb functional molecules on their outer surface or open channels and trap these molecules in the framework . In addition, functional molecules can be synthesized by one-pot method or post-synthetic modification, and can be incorporated into MOFs through covalent bonds. There are four main advanced strategies to functionalize MOFs with biological application therapeutics, including: surface adsorption, pore encapsulation, covalent bonding, and functional molecules as building blocks.
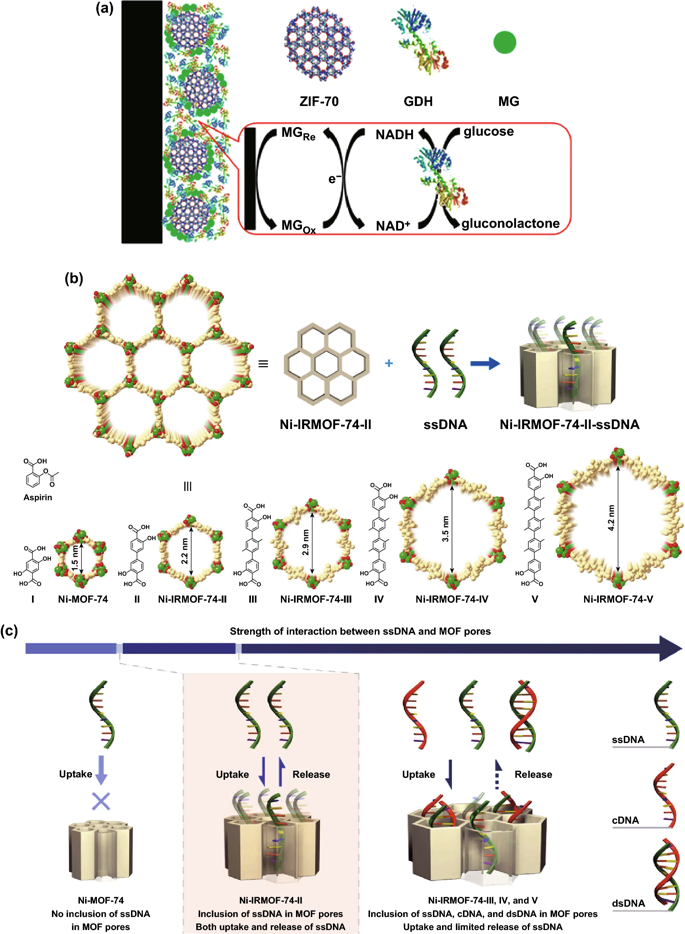
Figure 1. a The schematic diagram of preparing a glucose electrochemical biosensor using ZIF-70 as a substrate and co-immobilizing MG and GDH on the electrode surface. b Schematic diagram of ssDNA immobilized in Ni-IRMOF-74 series with precise control of channel size. Nickel, carbon and oxygen atoms are marked in green, gold and red respectively. c As the size of the MOF channel increases, the interaction between ssDNA and MOFs gradually increases. The relatively weak interaction ensures the absorption, protection and reversible release of ssDNA.
III. Application in drug delivery A major problem with conventional chemotherapy is that due to poor biodistribution, high doses of drugs are required, which leads to frequent dose-related side effects. This requires exploring new and efficient drug delivery systems (DDSs). Recent studies have shown that the application of MOF nanocarriers can achieve targeted drug delivery, increase cell uptake and control drug release, making MOFs a promising class of drug release DDS, including anti-cancer drugs, antibacterial drugs, metabolic marker molecules, Anti-glaucoma drugs and hormones.
3.1 Drugs
Generally, drugs are loaded into MOFs through in-situ encapsulation or post-synthesis modification strategies. The former is a relatively simple method, suitable for heat-stable drugs to overcome premature release of drugs. The latter provides a gentler environment to avoid destroying drug molecules. With the development of MOF chemistry, a series of MOFs have been explored as promising candidate carriers for application in this field.
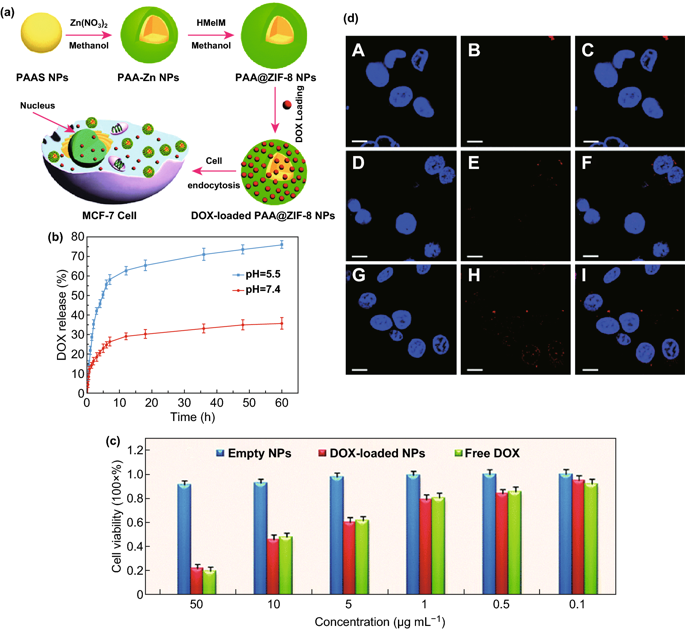
Figure 2. a Comprehensive schematic diagram of PAA@ZIF-8 as a nanocarrier for DOX drug loading and pH controlled release. b The drug release of DOX-loaded PAA@ZIF-8 at pH values of 5.5 and 7.4 and temperature of 37��C. c Toxicity of different concentrations of PAA@ZIF-8, load DOXPAA@ZIF, and single-drug DOX to MCF-7 cells after 24 h incubation. d MCF-7 cells uptake CLSM to cells loaded with DOX PAA@ZIF-8 ([DOX]=20 ��g/mL) incubated at 37�� for 3 hours (AC), 12 hours (DF) and 24 hours (gI) image. Columns 1-3 can be cell nucleus (Hoechst 33342 stained blue), DOX loaded PAA@ZIF-8, and the combined image of the two. The scale bar is 10 ��m.
3.2 Nucleic acid
Nucleic acids include deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Nucleic acid plays an important role in the storage and expression of genetic information. In general, the addition of nucleic acids to MOFs nanocarriers can prevent their degradation and accelerate their cellular uptake. In addition, surface modification of MOFs nanoparticles with nucleic acids can improve their colloidal stability by providing steric hindrance and static resistance aggregation. Currently, MOFs have been studied for DNA, small interfering RNA (siRNA), nucleic acid aptamer delivery or controlled release.
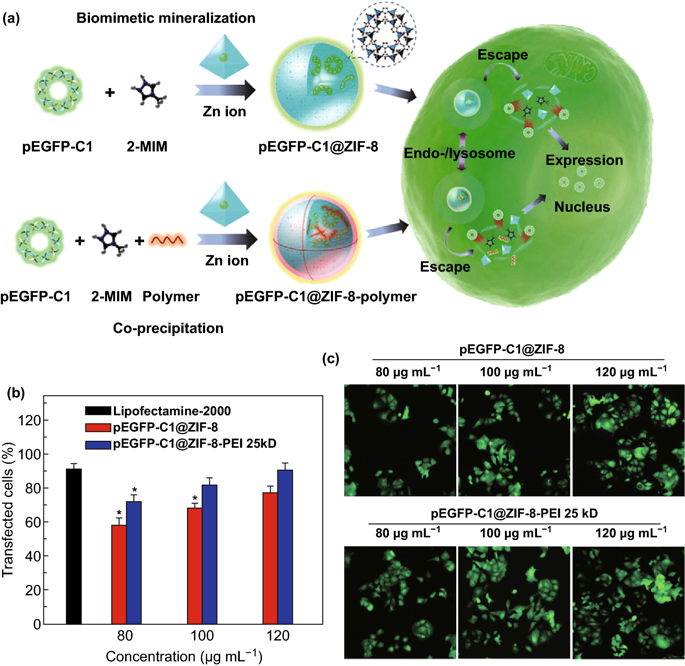
Figure 3. a schematic diagram of the cellular uptake and expression of pEGFP-C1@ZIF-8 through biomimetic mineralization and pEGFP-C1@ZIF-8-polymer through co-precipitation. b Transfection efficiency of pEGFP-C1@ZIF-8, pEGFP-C1@ZIF-8-PEI 25kD, lipofectamine-2000 at different concentrations. c A typical CLSM image of pEGFP-C1 expressed in MCF-7 cells.
3.3 Protein
Proteins have many functions, such as DNA replication, catalysis of metabolic reactions, and molecular transportation. Because proteins are bulky, charged on the surface, and sensitive to the environment, it is difficult for proteins to naturally pass through cell membranes without losing structural integrity. In order to use proteins for therapeutic purposes, MOFs nanoparticles for intracellular delivery of proteins have attracted more and more attention in recent years.
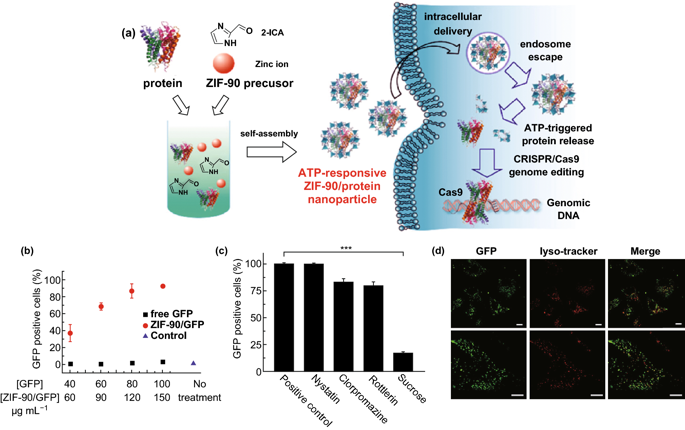
Figure 4. a Schematic diagram of the synthesis of ZIF-90/protein nanoparticles and ATP triggering protein release in cells. b Cellular uptake efficiency of ZIF-90/GFP. c The effect of different endocytosis inhibitors on the uptake efficiency of ZIF-90/GFP cells. d CLSM image of HeLa cells treated with ZIF-90/GFP. Inclusion body/lysosome staining was performed with lysosomal probe (red). IV Challenges Although significant achievements have been made in the application of MOFs in the field of drug delivery, there are still some challenges in this field: 1) So far, the research on drug loading and release kinetics of drugs is still very limited; 2) The potential toxicity of MOF-based DDS in clinical applications. The existing literature on the toxicity studies of MOF nanoparticles is very limited, and it is not enough to draw conclusions. In order to reach the clinical development stage of MOF nanoparticles, the preclinical evaluation performance of DDS based on MOFs should be optimized through systematic in vivo studies on its stability, degradation mechanism and side effects on normal organs.
Article source nanomicroletters nanomicroletters
This information is sourced from the Internet for academic exchange only. If there is any infringement, please contact us to delete it immediately.








