�Ѵ��ļ���photo/1772588079.png
Nanoplastic (NPs) pollution is one of the most enduring environmental crises of our time, requiring breakthrough material technologies for effective remediation. This study engineered semi-flexible micro-nanostructured N-modified cellulose nanofibers@layered double hydroxide (N-CNF@LDH) composite aerogels to achieve efficient removal of NPs through dynamic hydrogen bonding and coordination interactions, with LDH nanoflower clusters uniformly anchored on the nitrogen-modified cellulose honeycomb framework.Under the action of dynamic hydrogen bonds, the structure spontaneously reorganizes to a new equilibrium position after the external force is released, resulting in reversible opening and closing movements within the flower clusters. This design achieves ultra-high NP adsorption (3648 mg��g−1) through the reversible opening of channels between petals, while compression-triggered petal contraction enables instantaneous release (with a release rate of 96% within just 10 seconds), and the desorption kinetics are nearly three orders of magnitude faster than conventional adsorbents. In addition, the dynamic network achieves rapid structural reconstruction of N-CNF@LDH through hydrogen bond assembly, maintaining 80.4% NP capacity and 95.2% shape recovery after 100 cycles. It demonstrates outstanding NP removal performance in a variety of practical water systems while maintaining cost-effectiveness, breaking through the fundamental capacity-regeneration trade-off in environmental remediation.
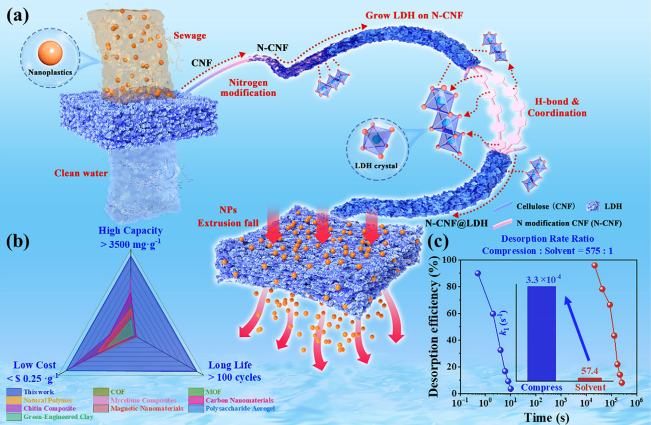
(a) Schematic diagram of N-CNF @LDH composite aerogel capturing nanoparticles and desorption induced by extrusion. (b) Comparison of the performance coefficients of N-CNF @LDH composite aerogel with other adsorbents reported in the literature. (c) Desorption rate of N-CNF @LDH composite aerogel within 10 seconds (schematic diagram comparing extrusion-driven desorption with desorption kinetics by conventional solvent immersion).
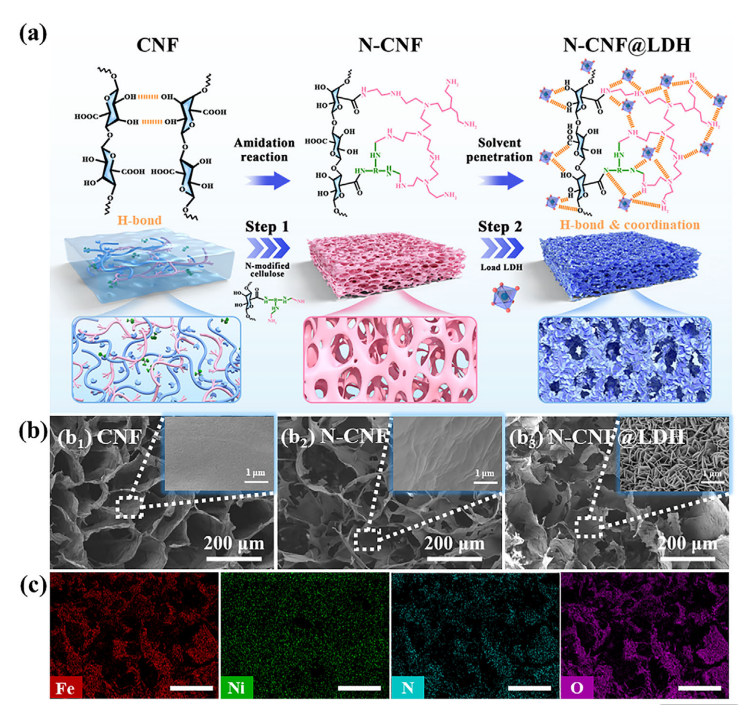
Synthesis and morphological characterization of N-CNF@LDH composite aerogel. (a) Schematic diagram of the preparation process of N-CNF@LDH. (b) Representative SEM images: b1) CNF, b2) N-CNF, b3) N-CNF@LDH. (c) Corresponding SEM-EDS elemental distribution maps of N-CNF@LDH (scale bar: 200 µm).
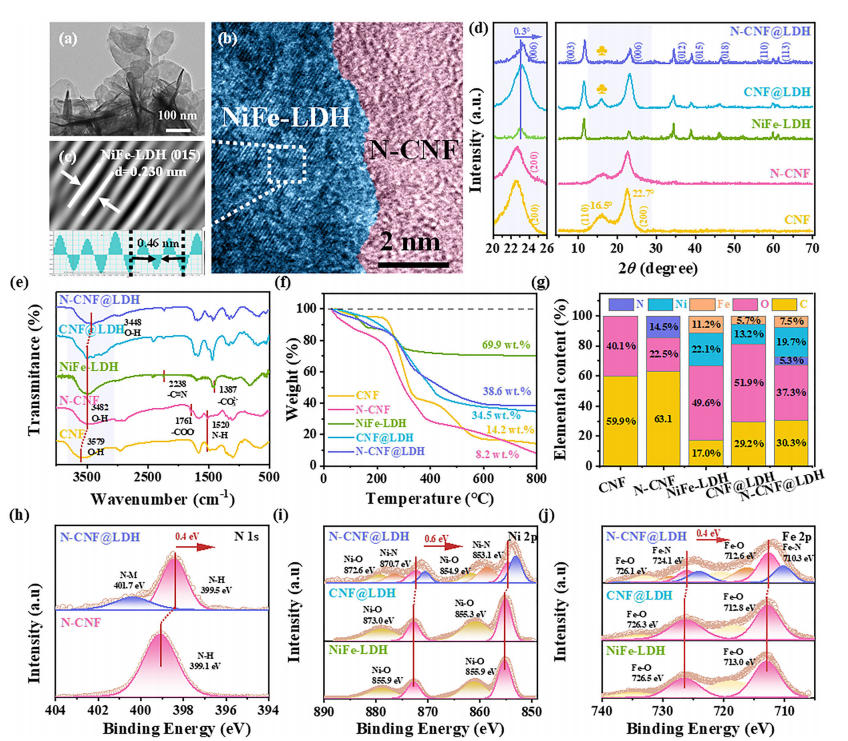
Composition and structural characterization of the samples. (a) TEM image of N-CNF@LDH, (b, c) HRTEM images, (d) XRD patterns of CNF, N-CNF, NiFe-LDH, CNF@LDH, and N-CNF@LDH, (e) FTIR spectra and (f) TGA curves. (g) Elemental composition ratio, (h) N1s high-resolution spectra of N-CNF and N-CNF@LDH, (i) Ni2p and (j) Fe2p high-resolution spectra of NiFe-LDH, CNF@LDH, and N-CNF@LDH.
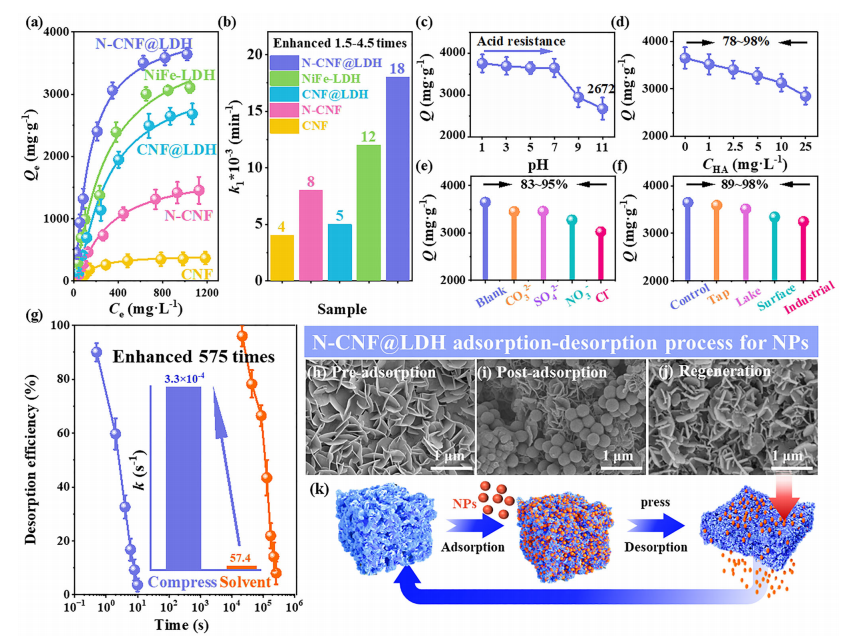
Evaluation of adsorption-desorption performance of nanoparticles. (a) Adsorption isotherms of nanoparticles on CNF, N-CNF, NiFe-LDH, CNF@LDH, and N-CNF@LDH, and (b) kinetics. (c) Effect of different pH values, (d) HA concentration, and (e) types of ions on the nanoparticle adsorption performance of N-CNF@LDH. (f) Effect of N-CNF@LDH on nanoparticle adsorption capacity in different actual water environments. (g) Desorption rate of N-CNF@LDH within 10 seconds (schematic comparing compression-driven desorption and desorption kinetics by traditional solvent immersion). (h�Cj) Time-dependent SEM images of the N-CNF@LDH desorption process. (k) Schematic of N-CNF@LDH adsorption-desorption.
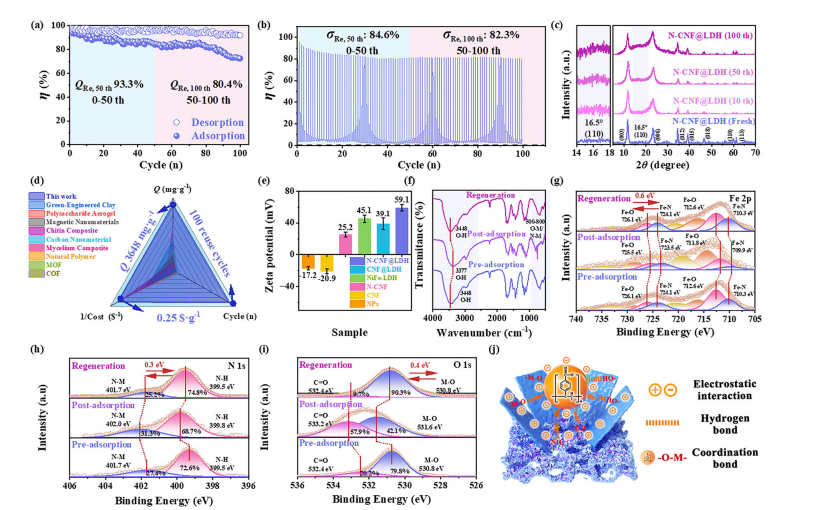
Cyclic stability, structural characterization, and nanoparticle removal mechanism of N-CNF@LDH aerogel. (a) Adsorption-desorption cycle performance of N-CNF@LDH for nanoparticles over 100 cycles (initial Q value of 3648 mg⋅g−1) and (b) stress-strain curves during this cycle (�� represents stress, maximum stress 130 kPa). (c) XRD pattern of regenerated N-CNF@LDH. (d) Radar chart comparing the overall performance (adsorption capacity, cyclic stability, and economic cost) of N-CNF@LDH with reported adsorbents. (e) Zeta potential comparison of NiFe-LDH, CNF, N-CNF, and N-CNF@LDH composites. (f) FTIR spectra of N-CNF@LDH before and after nanoparticle adsorption. (g-i) High-resolution XPS spectra: (g) O 1s, (h) N 1s, and (i) Fe 2p fine spectra. (j) Schematic diagram of the synergistic mechanism of nanoparticle removal by N-CNF@LDH.
Summary
A study published in Advanced Functional Materials reported a semi-flexible micro-nano composite aerogel (N-CNF@LDH) based on dynamic hydrogen bonding and coordination interactions, designed for the efficient removal and rapid regeneration of nanoplastics in water. The research team constructed the composite aerogel with a dynamic reversible structure by in situ growing nickel-iron layered double hydroxide (NiFe-LDH) nanoflower clusters on the surface of nitrogen-modified cellulose nanofibers (N-CNF). This material leverages the synergistic coordination and hydrogen bonding between the metal active sites on the LDH surface (Ni²⁺/Fe³⁺) and the amino/hydroxyl groups on the N-CNF surface, forming a reversible "petal-channel" opening and closing mechanism during the adsorption process. This continuously exposes fresh adsorption sites, significantly enhancing the adsorption capacity for polystyrene nanoplastics, reaching 3648 mg��g⁻¹.
Reference News:
DOI: 10.1002/adfm.202526566








