
Graphyne (GDY) has rich carbon chemical bonds, high π conjugation, wide interplanar spacing, uniformly dispersed channel configuration, natural band gap, excellent chemical and thermal stability, and is known as the most stable artificial Allomorphs of synthetic diacetylene carbons. It shows great application potential in the fields of new energy, catalysis and photoelectric conversion. The theory predicts that as the dimension decreases, the performance of graphyne is greatly regulated. The band structure of one-dimensional graphynes is related to the edge structure and bandwidth. The band gap of graphynes decreases as the width of the nanostrips increases. Therefore, graphynes have important significance in the study of nanoscale electronic devices. There are two ways to synthesize graphite alkyne nanometers, one is the "top-down" method and the other is the "bottom-up" method. The "top-down" idea, such as cutting from graphyne, will cause the width and edge structure of the prepared graphyne nanoribbons to be uncertain. The "bottom-up" chemical synthesis method can produce graphitic nanoribbons with uniform structure and uniform width. However, the selective and gradual coupling of acetylene groups must be controlled during the synthesis process, and the synthesis of graphene acetylene nanoribbons from the "bottom-up" method is still a challenge.
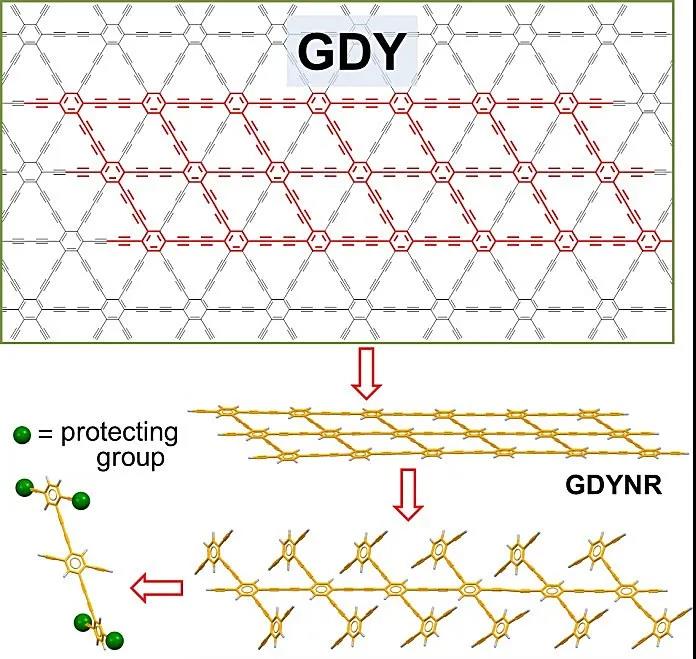
Figure 1. Schematic and reverse synthesis analysis of graphyne.
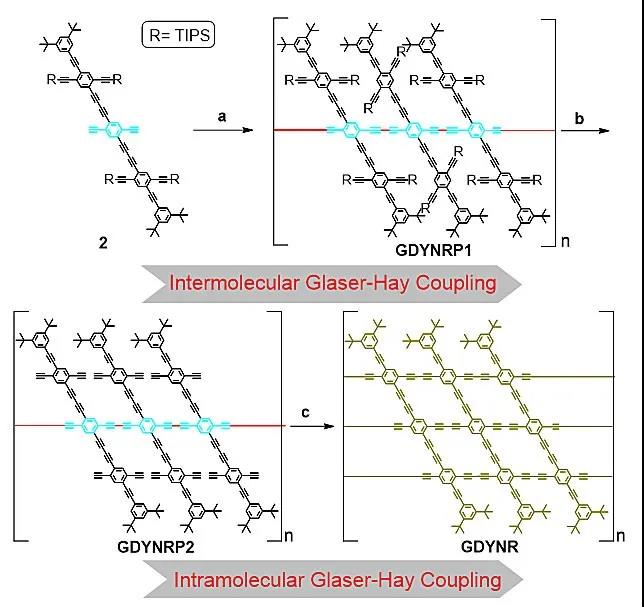
Figure 2. Step-by-step intermolecular and intramolecular Glazer-Hay coupling reactions for the synthesis of graphynyne nanoribbons.
Recently, Academician Li Yuliang and Researcher Li Yongjun (corresponding author) of the Institute of Chemistry of the Chinese Academy of Sciences reported a two-step method for the preparation of graphene alkyne nanoribbons. As shown in the reverse synthesis analysis shown in FIG. 1, the polymerization of the acetylene group at the center of the monomer is first performed to ensure the one-dimensional structure of the graphyne nanoribbons. Then the intramolecular coupling of ethynyl groups on adjacent side chains is performed with the aid of the liquid-liquid interface. The steric hindrance provided by the large volume of 3,5-di-tert-butylphenyl on the edge can further reduce the molecule Coupling. The first chemically synthesized graphynyne nanoribbons (Figure 1) were successfully prepared through the above strategy. It consists of a benzene ring and a butadiynyl rhombus. . The authors also studied the assembly of graphene acetylene nanoribbons, which can be woven into nano-fabric thin films through π-π interactions, which can be used to construct protective films for lithium electrodes in lithium-ion batteries, which can effectively inhibit the formation of lithium dendrites and improve lithium Ion battery stability.
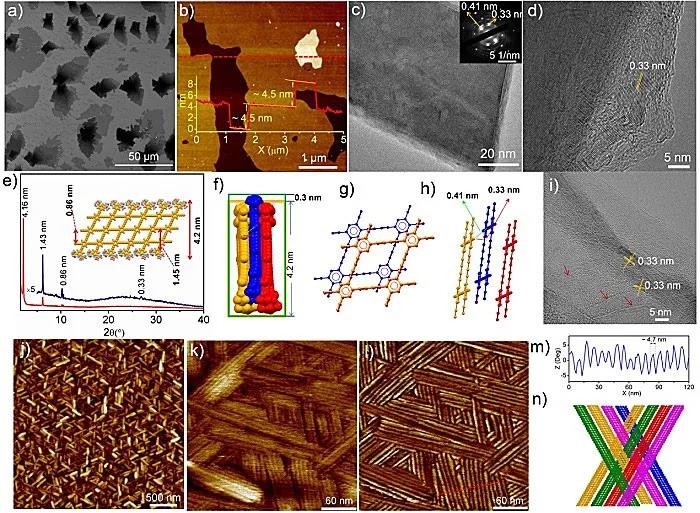
Figure 3. Structural characterization and assembly behavior of graphyne ribbons.
This "bottom-up" synthesis method is also applicable to the preparation of other graphene alkyne nanoribbons with different widths and different edge structures, such as concave or serrated. In addition, by grafting different functional groups on the outer edges of the nanoribbons, the electrical and optical properties of the graphyne nanoribbons can be regulated, and further potential applications in the fields of electronic devices, energy conversion, and energy storage have been developed.
Controllable Synthesis of Graphdiyne NanoribbonsWeixiang Zhou, Han Shen, Yan Zeng, Yuanping Yi, Zicheng Zuo, Yongjun Li, Yuliang LiAngew. Chem. Int. Ed., 2020, DOI: 10.1002/anie.201916518







