ORR (oxygen reduction reaction), OER (oxygen evolution reaction) and HER (hydrogen evolution reaction) are three key reactions involved in new energy technologies such as metal-air batteries, electrocatalytic water decomposition devices, etc., but due to their slow reaction kinetics, the reactions High overpotentials are usually required to drive. Therefore, it is very important and very challenging to build an efficient three-function electrocatalyst for HER, OER and ORR. Among many compounds, MoS₂ is a promising HER electrocatalyst, but its poor conductivity and strong agglomeration during the preparation process have severely restricted its catalytic effect. In contrast, Co₉S₈ has attracted wide attention due to its relatively low electrical conductivity and good OER electrocatalytic performance. Although these two compounds have their own advantages, they are limited by a single catalytic function, and it is difficult to expand their application range. An effective synthesis strategy is urgently needed, which can combine the unique advantages of each component to create a rich interface and shorten the ion diffusion path. And reduce the interface contact resistance, thereby building a multifunctional nanocatalyst with high catalytic activity. The development and construction of this new type of nanostructure and the development of multifunctional electrocatalysts are of great significance, but also very challenging.
ORR (oxygen reduction reaction), OER (oxygen evolution reaction) and HER (hydrogen evolution reaction) are three key reactions involved in new energy technologies such as metal-air batteries, electrocatalytic water decomposition devices, etc., but due to their slow reaction kinetics, the reactions High overpotentials are usually required to drive. Therefore, it is very important and very challenging to build an efficient three-function electrocatalyst for HER, OER and ORR. Among many compounds, MoS₂ is a promising HER electrocatalyst, but its poor conductivity and strong agglomeration during the preparation process have severely restricted its catalytic effect. In contrast, Co₉S₈ has attracted wide attention due to its relatively low electrical conductivity and good OER electrocatalytic performance. Although these two compounds have their own advantages, they are limited by a single catalytic function, and it is difficult to expand their application range. An effective synthesis strategy is urgently needed, which can combine the unique advantages of each component to create a rich interface and shorten the ion diffusion path. And reduce the interface contact resistance, thereby building a multifunctional nanocatalyst with high catalytic activity. The development and construction of this new type of nanostructure and the development of multifunctional electrocatalysts are of great significance, but also very challenging.
Recently, the research group of Professor Li Liping from the State Key Laboratory of Inorganic Synthesis and Preparation of Jilin University successfully used the metal-organic framework ZIF-67 as a precursor to successfully prepare a unique core-double shell nanostructured compound Co₉S₈ @ Co₉S₈ @ MoS₂-0.5. The unique synthesis strategy not only enables the nanostructures to inherit the complete morphology of the precursors, but also produces a rich interface to effectively regulate the electronic structure of each component. Co₉S₈ @ Co₉S₈ @ MoS₂-0.5, which has a novel core-double shell nanostructure, shows excellent HER, OER, and ORR electrocatalytic activity.
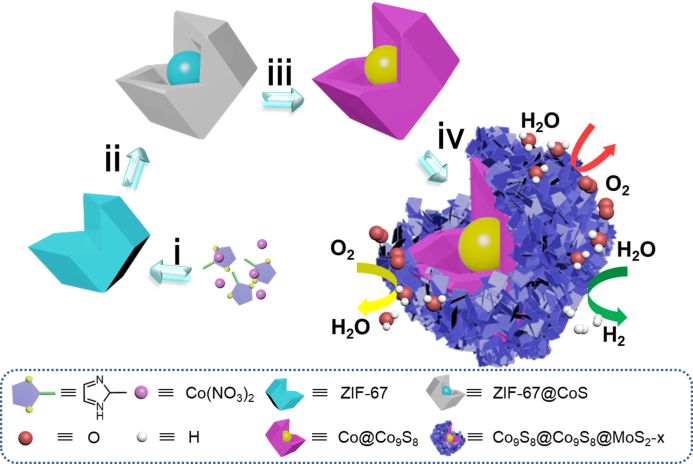
Figure 1. Schematic of Co9S8 @ Co9S8 @ MoS2-x synthesis
The preparation process of Co₉S₈ @ Co₉S₈ @ MoS₂-0.5 is shown in Figure 1: First, the ZIF-67 precursor is grown at room temperature (step i); in order to obtain the core-shell structure intermediate, ZIF-67 and thioethyl ZIF-67 @ CoS with core-shell structure is obtained by refluxing amide (TAA) in ethanol solution (step ii); then ZIF-67 @ CoS is further calcined in an inert atmosphere at high temperature to form Co @ Co₉S₈, in which ZIF-67 is cored. The carbonization reduction is called the metallic cobalt phase, and the shell CoS phase becomes Co₉S₈ (step iii); the next step is to grow MoS₂ on Co @ Co₉S₈ under hydrothermal conditions and heat treatment to increase the degree of crystallization of the material. Adjusting the amount of molybdate can adjust the relative content of Co₉S₈ and MoS₂ (step iv) to optimize the catalytic ability of the material.
Figure 2. SEM, TEM, HRTEM, and EDX spectra of Co9S8 @ Co9S8 @ MoS2-0.5.
It can be seen from the SEM image that Co₉S₈ @ Co₉S₈ @ MoS₂-0.5 still maintains a regular dodecahedron structure similar to that of the precursor ZIF-67. From the TEM image, it can be seen that this unique core-double shell structure has a hollow interior and a rough outer surface. HRTEM can distinguish the crystal planes of Co₉S₈ (200) and MoS₂ (002). The high-angle annular dark field shows its unique core-double shell structure. The elements Co, S and Mo are evenly distributed in the Co₉S₈ @ Co₉S₈ @ MoS₂-0.5 structure.
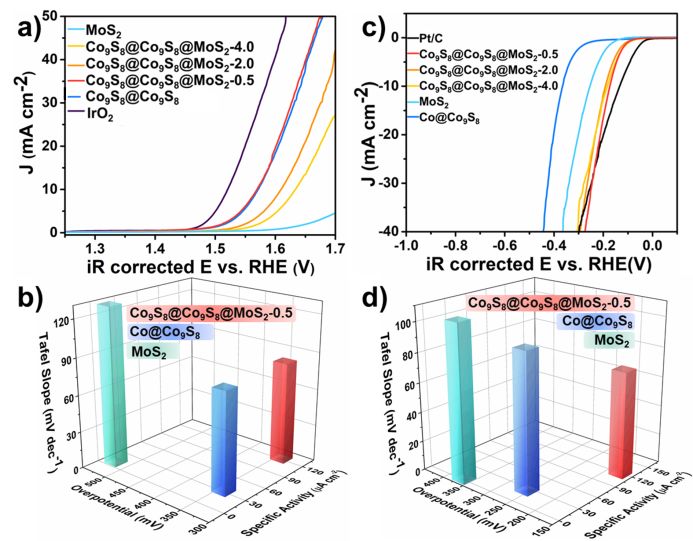
Figure 3. Electrochemical properties of Co9S8 @ Co9S8 @ MoS2-x compounds at different ratios a) OER polarization curve; b) Comparison of Tafel slope, overpotential, and specific activity compared to the catalytic OER process of different compounds; c) HER Polarization curve; d) Comparison of Tafel slope, overpotential and specific activity of catalytic HER process comparing different compounds
By evaluating the electrochemical activity of the materials, we found that Co₉S₈ @ Co₉S₈ @ MoS₂-0.5 exhibits excellent catalytic performance in the process of electrocatalytic OER, requiring only 340 mV overpotential to achieve a current value of 10 mA cm⁻2. In addition, when catalyzing HER, Co₈S₈ @ Co₉S₈ @ MoS₂-0.5 shows better catalytic performance than single-phase MoS₂ and Co₉S₈. It only needs 170 mV overpotential drive at a current of 10 mA cm⁻2.
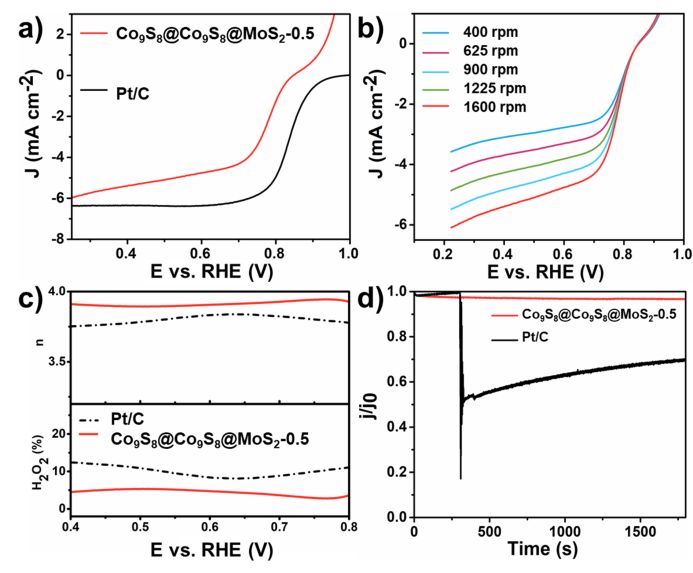
Figure 4. Electrochemical performance curves of Co9S8 @ Co9S8 @ MoS2-x and Pt / C catalyzed ORR a) Linear scanning voltammetry diagram; b) Linear scanning voltammetry diagram at different speeds; c) Hydrogen peroxide and electron transfer number D) Methanol cross tolerance test
It is very interesting that with this unique core-double shell structure Co₉S₈ @ Co₉S₈ @ MoS₂-0.5, it can also show good electrocatalytic ORR capability. The limit diffusion current reaches 0.66 mA cm⁻2 at 0.6 V (vs. RHE), and the half-wave potential is about 0.776 V (vs. RHE). By rotating the ring disk electrode, it can be confirmed that the number of electron transfer during the reaction is close to 4, which proves that the catalytic reaction passes through a four-electron path. Not only that, the material can show good methanol resistance in the methanol cross-resistance test.
This work provides an innovative perspective for the synthesis of new heterostructure materials and a feasible strategy for exploring the synthesis of multifunctional electrocatalysts, which is of great significance for the development of MOF-based multifunctional catalyst design methods.
norg. Chem. Front., 2020,7, 191-197
https://doi.org/10.1039/C9QI01080G
Source: Frontiers Journals